HOME PRODUCTS LAB DIVISION
The FOB4 Stand-alone Lab Sterilizer is an autoclave that combines excellent performances with overall high quality materials and ease of use.
The FOB4 Lab Sterilizer is suitable for the use in laboratory for many different applications, such as:
- Microbiology and analytical labs.
- Research institutes and examination agencies.
- Bio-Technologies and life sciences.
- Animal facility and clinical diagnostic labs.
- Pharmaceutical, food, chemistry industries, QC labs.
- Agricultural, environmental and veterinary labs.
These sterilizers have been specially designed with ergonomical features for easy handling of higher volume sterilization materials and goods as: glassware, media preparation, liquids, in open and sealed containers, mixed regular and high risk waste, as well as surgery instruments, textiles, rubber articles and packaged tools.
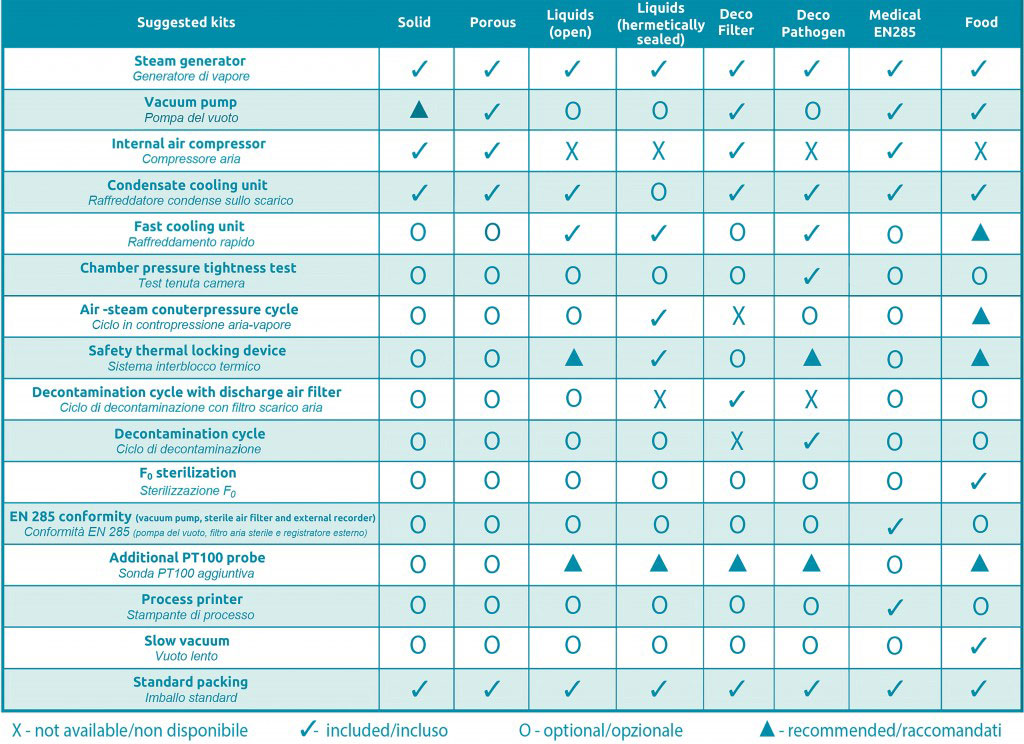
FOB4 Stand-alone Lab Sterilizer is easy to be configured, thanks to its conceptual design and modular construction with a convenient portfolio of optional kits. In fact, all the models can be configured using various customized systems guaranteed by the wide range of processes, software options and an extensive choice of accessories. Please, click on the link “recommended kit” to check out all possible configurations referred to FOB4 series.
DESIGN AND TECHNICAL FEATURES
FOB4 can be designed in two chamber dimensions: 147 liters (FOB4-TS) with single vertical sliding door or 210 liters (FOB4S-TS – FOB4/L TS), 226 liters (FOB4L) with double vertical sliding door for pass-through purposes. The vertically automatic sliding doors are pneumatically activated for space efficiency in laboratory and hospitals.
FOB4 autoclave has internal 316L stainless steel plates fed by steam used both as heat-exchanger system for steam pre-heating and to cool the chamber temperature by means of cold softened water. As an option these plates can be used for drying purposes at the end of the cycle.
Fedegari designs laboratory autoclaves combining operators’ safety as well as equipment performances. Thermal blocking system to prevent door openings in hazardous situations. Temperature probes and pressure transducers ensure constant process monitoring, as well as precision and optimal temperature distribution in the chamber, guaranteeing a high level of safety for the operator and performance and reproducibility of the sterilization process.
FOB series is equipped with DCS20 process controller: 30 cycles easy to customize in a multi-user environment. Large touch-screen color display and interface for remote monitoring via Ethernet control. DCS20 process controller is fully validated and documented.
KEY BENEFITS
- Energy and water cost-saving.
- Small foot print with superior loading capacity.
- Ergonomic positioning of adjustable operator display.
- Full and easy accessibility to technical area from front and lateral sides.
- Special test programs for routine check of sterilizer efficiency.
- GAMP5 compliant.
- GLP compliant.
- Reduced process time.
- Flexible process machine
COMPLIANCE TO
- PED Directive 2014/68/UE – Pressure equipment
- Machine Directive Macchine 2006/42/EC
- EMC Directive 2014/35/UE
- LVD Directive 2006/95/UE
Want to know more about our certifications? Please visit the dedicated section or do not hesitate to contact us.
STANDARD
- EN ISO 12100 Safety of machinery – General principles for design – Risk assessment and risk reduction
- EN ISO 13857 Safety of machinery – Safety distances to prevent hazard zones being reached by upper and lower limbs
- EN ISO 13849-1 Safety of machinery – Safety-related parts of control systems – Part 1: General principles for design
- EN IEC 60204-1 Safety of machinery – Electrical equipment of machines – Part 1: General requirements
- EN IEC 61326-1 Electrical equipment for measurement, control and laboratory use – EMC requirements – Part 1: General requirements
- EN 61010-1 Safety requirements for electrical equipment for measurement, control, and laboratory use – Part 1: General requirements
- EN 61010-2-040 Safety requirements for electrical equipment for measurement, control, and laboratory use – Part 2-040 Particular requirements for sterilizers and washer-disinfectors used to treat medical materials
- EN 13445-3 Unfired pressure vessels – Part 3: Design
- EN 4126-1 Safety devices for protection against excessive pressure – Part 1: Safety valves
STANDARD COMPLIANCE FOR NON-EU COUNTRIES
United States of America (USA)
- ASME (American Society of Mechanical Engineers) U & S Stamp
- UL 508 A Standard for Industrial Control Equipment
- NPFA 70 National Electrical Code (NEC
- NPFA 79 Electrical Standard for Industrial Machinery
People’s Republic of China
- SELO – Chinese Safety Regulation for Pressure Vessel
Russian Federation
- TR CU 032 On the safety of equipment operating under excessive pressure
- TR CU 010 Machinery Directive
Brazil
- NR 13 Boilers and Pressure Vessel
Malaysia
- Department of Occupational Safety and Health (DOSH)
Singapore
- Ministry of Manpower (MOM)
DOWNLOAD
RELATED PRODUCTS
DCS20
The DCS20 process controller was specially designed for laboratory machines manufactured by Fedegari.
The result is a modular system that offers a wide range of functions and high reliability.
The system includes a supervisor module and other individual ones dedicated to specific functions such as: programs management, cycles, phases, configurations, and alarms for the module.
Options
- REMOTE GUI allows remote connection to a PC by Ethernet connection. This connection allows the operator to view the machine display on the desktop.
- REMOTE CONTROL allows remote connection to a PC by the Ethernet connection port. This connection allows the operator to fully interact with the machine. This excludes door closing and data backup operations.
Data management in compliance with the requirements of the 21 CFR PART 11 protocol.
Life cycle and Validation
DCS20 is designed, developed, tested, maintained, and validated according to a defined life cycle according to regulatory requirements stated in the current GMP.
Life cycle management complies with FDA 21 CFR Part 11, up to the current operational stage in defining product management practices in accordance with the principles of the GAMP5 guideline.
According to the system classification defined in GAMP 5 – Appendix M4 Categories of Software and Hardware, DCSPLUS20 is classified as:
- Software Category 4 – Configurable system
- Hardware Category 1 – Standard hardware components
Tests on software and hardware components are performed, during project and engineering activities, by applying functional risk analysis according to ALCOA+ principles, as defined in the Change Control procedure, dedicated to DCS20, part of the Fedegari Quality System.