HOME PRODUCTS LAB DIVISION
FOB is a benchtop lab sterilizer that combines excellent performance with high quality materials, ease of use and reduced utilies consumption.
The FOB series are suitable for many different applications, making it the perfect choice to satisfy modern laboratory needs. FOB series autoclaves combines technology and know-how, and can be used by very different laboratories, such as:
- Private and public microbiology labs.
- High schools and Universities.
- Research institutes, life sciences and biotech labs.
- Clinical diagnostic labs.
- Agriculture, environmental and veterinary labs.
- Material testing laboratories.
- Quality control lab´s in pharma, food/beverage, chemistry/cosmetics and other industrial sectors.
FOB may be adapted to satisfy various laboratory applications and the treatment of many different types of loads, including waste with high pathogen risk, in addition to solid porous goods, packaged items and liquid/media, in open and sealed containers.
FOB series autoclaves are used in highly specialized fields thanks to a wide selection of options on a modular system. Hence, FOB sterilizers can be configured to meet the most demanding user’s requirements.
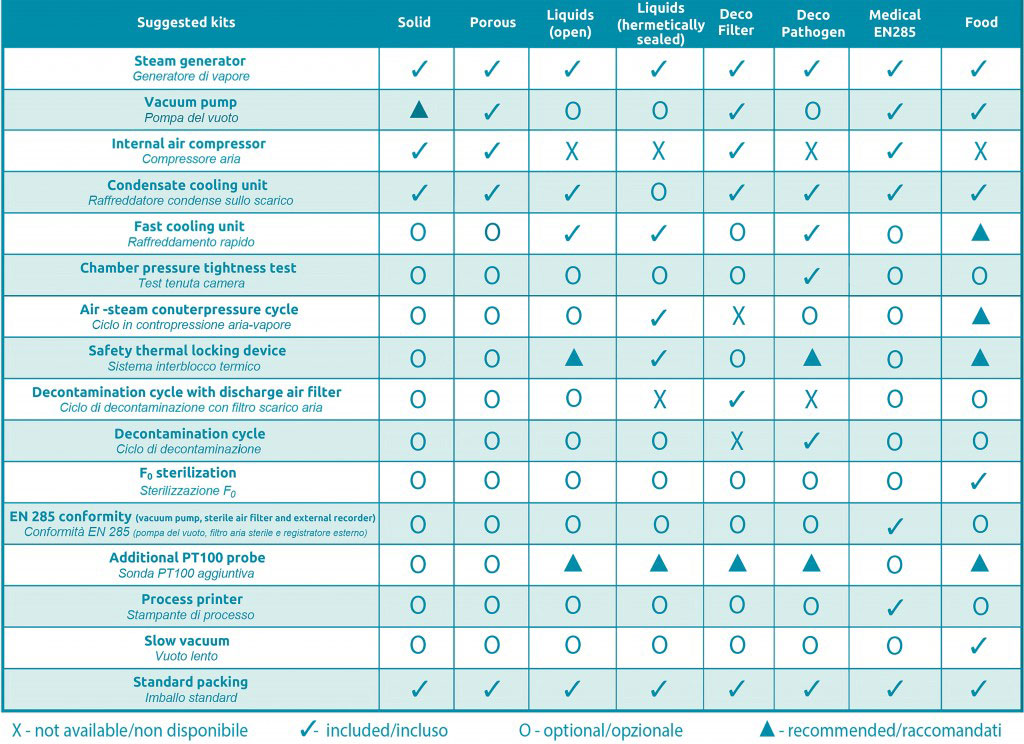
Please, click on the link “recommended kit” to check out all possible configurations referred to FOB series.
DESIGN AND TECHNICAL FEATURES
FOB series of bench top lab sterilizers can be designed with four different chamber dimensions with single or double hinged doors for pass-through applications:
- FOB2-TS: 47 liters, single hinged door.
- FOB2S-TS: 75 liters, double hinged door.
- FOB3-TS: 90 liters, single hinged door.
- FOB3S-TS: 122 liters, double hinged door.
A unique opening/closing door system with patented pneumatic gasket seal guarantees perfect airtight, maximum reliability and improved safety for operators. Moreover, FOB autoclaves can be equipped with safety device to prevent door opening.
Chambers, pneumatic valves and hydraulic components are made of 316L stainless steel, with sanitary electropolished finishing. FOB is designed to assure wide and easy access to technical area for maintenance activities.
Easy installation and quick connection to the utilities makes FOB series autoclave even more attractive. To assure energy savings, FOB has a heat exchanger recovery system installed out of the chamber to reuse the heat of condensate for water pre-heating before the steam generation.
FOB series is equipped with DCS20 process controller: 30 cycles easy to customize in a multi-user environment. Large touch-screen color display and interface for remote monitoring via Ethernet control. DCS20 process controller is fully validated and documented.
KEY BENEFITS
- Modular concept: extremely flexible configuration.
- Energy saving system.
- Increase of the maximum temperature and the pressure
- Adjustable operator display for an ergonomic postioning.
- Space optimization: load capacity improved
- Extensive choice of loading equipment (racks, adjustable shelves, baskets, etc.).
- High level of programming with 30 configurable cycles .
- Special test programs (i.e. Bowie-Dick, vacuum, pressure leak, etc.) for routine check of sterilizer efficiency.
- GLP compliance
COMPLIANCE TO
- PED Directive 2014/68/UE – Pressure equipment
- Machine Directive Macchine 2006/42/EC
- EMC Directive 2014/35/UE
- LVD Directive 2006/95/UE
Want to know more about our certifications? Please visit the dedicated section or do not hesitate to contact us.
STANDARD
- EN ISO 12100 Safety of machinery – General principles for design – Risk assessment and risk reduction
- EN ISO 13857 Safety of machinery – Safety distances to prevent hazard zones being reached by upper and lower limbs
- EN ISO 13849-1 Safety of machinery – Safety-related parts of control systems – Part 1: General principles for design
- EN IEC 60204-1 Safety of machinery – Electrical equipment of machines – Part 1: General requirements
- EN IEC 61326-1 Electrical equipment for measurement, control and laboratory use – EMC requirements – Part 1: General requirements
- EN 61010-1 Safety requirements for electrical equipment for measurement, control, and laboratory use – Part 1: General requirements
- EN 61010-2-040 Safety requirements for electrical equipment for measurement, control, and laboratory use – Part 2-040 Particular requirements for sterilizers and washer-disinfectors used to treat medical materials
- EN 13445-3 Unfired pressure vessels – Part 3: Design
- EN 4126-1 Safety devices for protection against excessive pressure – Part 1: Safety valves
Standard compliance for non-EU countries
United States of America (USA)
- ASME (American Society of Mechanical Engineers) U & S Stamp
- UL 508 A Standard for Industrial Control Equipment
- NPFA 70 National Electrical Code (NEC
- NPFA 79 Electrical Standard for Industrial Machinery
People’s Republic of China
- SELO – Chinese Safety Regulation for Pressure Vessel
Russian Federation
- TR CU 032 On the safety of equipment operating under excessive pressure
- TR CU 010 Machinery Directive
Brazil
- NR 13 Boilers and Pressure Vessel
Malaysia
- Department of Occupational Safety and Health (DOSH)
Singapore
Ministry of Manpower (MOM)
DOWNLOAD
Catalog
FOB Benchtop Lab Sterilizer
White papers
Moist-heat sterilization – Highlights on equivalent time F0
RELATED PRODUCTS
DCS20
The DCS20 process controller was specially designed for laboratory machines manufactured by Fedegari.
The result is a modular system that offers a wide range of functions and high reliability.
The system includes a supervisor module and other individual ones dedicated to specific functions such as: programs management, cycles, phases, configurations, and alarms for the module.
Options
- REMOTE GUI allows remote connection to a PC by Ethernet connection. This connection allows the operator to view the machine display on the desktop.
- REMOTE CONTROL allows remote connection to a PC by the Ethernet connection port. This connection allows the operator to fully interact with the machine. This excludes door closing and data backup operations.
Data management in compliance with the requirements of the 21 CFR PART 11 protocol.
Life cycle and Validation
DCS20 is designed, developed, tested, maintained, and validated according to a defined life cycle according to regulatory requirements stated in the current GMP.
Life cycle management complies with FDA 21 CFR Part 11, up to the current operational stage in defining product management practices in accordance with the principles of the GAMP5 guideline.
According to the system classification defined in GAMP 5 – Appendix M4 Categories of Software and Hardware, DCSPLUS20 is classified as:
- Software Category 4 – Configurable system
- Hardware Category 1 – Standard hardware components
Tests on software and hardware components are performed, during project and engineering activities, by applying functional risk analysis according to ALCOA+ principles, as defined in the Change Control procedure, dedicated to DCS20, part of the Fedegari Quality System.